Summary
IPG11406 is a pioneering, first-in-class, orally administered EBI2 (GPR183) antagonist poised to transform the treatment paradigm for autoimmune diseases. Unlike current standard-of-care therapies such as corticosteroids and TNF-α inhibitors, which only provide symptomatic relief, IPG11406 targets the underlying pathogenesis of autoimmune disorders. It is designed to eliminate pathological immune cells, leading to complete resolution of inflammation.
With its novel mechanism of action, status as the only EBI2-targeting therapy in clinical development worldwide, and rapid, profound efficacy demonstrated in early clinical studies, IPG11406 is positioned to become a foundational disease-modifying therapy for a broad range of autoimmune conditions.
Mechanism of Action
EBI2 (also known as GPR183) is a G protein-coupled receptor that is overexpressed on a range of key pathological immune cells, including B cells, T cells, and macrophages. In multiple autoimmune diseases, EBI2 functions as a "cellular navigation system", directing these immune cells to migrate into target tissues, thereby initiating and amplifying inflammation and tissue damage.
As a potent, selective small-molecule antagonist, IPG11406 is specifically designed to block the EBI2-mediated signaling pathways. By inhibiting this pathway, IPG11406 effectively prevents the migration and proliferation of pathological immune cells. This action clears inflammatory cells from disease lesions, reduces the expression of pro-inflammatory cytokines, and ultimately attenuates disease progression by addressing the root cause of immune dysregulation.

Key Differentiation
■ Global First-in-Class Advantage: IPG11406 is the only EBI2 antagonist to have advanced to clinical trials worldwide, conferring a significant first-mover advantage in this untapped therapeutic space.
■ Disease-Modifying Potential: By targeting the etiology of autoimmune diseases rather than just symptoms, IPG11406 has the potential to induce long-term, treatment-free remission and reduce relapse rates.
■ Oral Administration Convenience: As a small-molecule drug, IPG11406 offers the convenience of oral dosing, significantly improving patient compliance and accessibility compared to injectable biologics, while also potentially reducing overall treatment costs.
■ Rapid Onset of Action: Preliminary clinical data in lupus nephritis (LN) patients demonstrate substantial improvements as early as two weeks of treatment. Notably, after only 28 days of therapy, proteinuria levels declined by up to 73.6% from baseline—a stark contrast to competing therapies that typically require months to show measurable efficacy.
■ Exceptional Safety Profile: Preclinical and early clinical data indicate that IPG11406 is well-tolerated, with a therapeutic window exceeding 150-fold the effective dose, distinguishing it from conventional immunosuppressants with narrow safety margins.
Clinical Trials Summary
■ Phase 1 (Completed): A randomized, double-blind, placebo-controlled study in healthy adult volunteers in China was successfully completed in March 2025. The trial confirmed the safety and tolerability of orally administered IPG11406 and established a favorable pharmacokinetic (PK) profile supporting once- or twice-daily dosing regimens.
■ Phase 1b/2a (2a Ongoing): A multi-center, multiple-dose clinical trial in patients with lupus nephritis was initiated in China in December 2024. Preliminary results to date have been highly encouraging: As of March 2026, twenty-three lupus nephritis patients have been enrolled. Among these patients, twenty of them have completed the 58-day treatment and observation. The preliminary results showed that 65% of patients (thirteen out of twenty) exhibited substantial (over 30%) reduction of 24-hour proteinuria, including 4 patients with complete renal response (CRR) and 2 patients with partial renal response (PRR). Patient stratification analysis revealed that patients who underwent renal biopsy within 2 years confirming pathological class of III/IV ± V exhibited 50% of CRR ( two out of four), suggesting that IPG11406 would achieve significantly higher CRR rate than that of the existing monoclonal antibody therapies which required renal biopsy within 6 months confirming active pathological class of III/IV ± V to achieve a therapeutic benefit in their 2-year clinical trials.

IPG11406 Ph2a: 24-hour proteinuria in lupus nephritis patients showed a significant reduction from baseline after treatment
Current Status
The Phase 1b/2a clinical trial of IPG11406 in lupus nephritis is actively progressing in China. We anticipate completing this trial by Q2 2026 and subsequently advancing to a pivotal Phase 2b study. In addition, we have successfully held the initiation meeting for the Phase 2 clinical trial in ulcerative colitis (UC), with Peking Union Medical College Hospital serving as the leading site. Ethics committee submissions and Human Genetic Resources Administration filings are being finalized, with the first patient expected to be enrolled in mid-2026.
Preclinical In vivo Efficacy
In comprehensive preclinical studies, IPG11406 has demonstrated robust, dose-dependent therapeutic efficacy across multiple validated animal models of major autoimmune diseases:
• Systemic Lupus Erythematosus/Lupus Nephritis (SLE/LN): In the MRL/lpr mouse model, IPG11406 treatment resulted in a significant, dose-dependent reductions in proteinuria, a key marker of kidney damage. It also markedly decreased inflammatory cell infiltration in the kidney, leading to a significant amelioration of glomerular and tubulointerstitial damage.
• Inflammatory Bowel Disease (IBD): In the Il10 knockout mouse model, IPG11406 produced a dose-dependent reduction in inflammatory cell infiltration in the colon and significant improvement in the overall colonic architecture, demonstrating its potential to control gut inflammation.
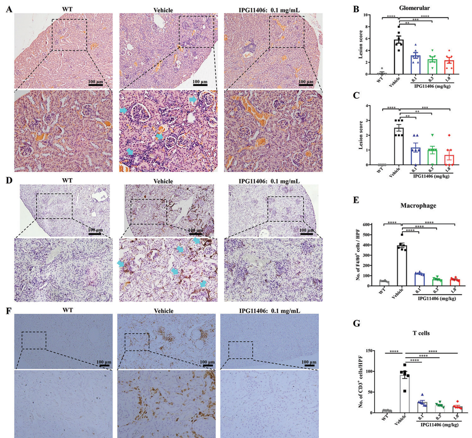
The changes in histological pathogenesis and inflammatory cell infiltration of the kidney in MRL/lpr mice.

Effects of IPG11406 on histopathological score of colon tissue in mice. (A) Inflammatory cell infiltration; (B) Histopathology score.
• Multiple Sclerosis (MS): In the experimental autoimmune encephalomyelitis (EAE) mouse model, IPG11406 treatment delayed disease onset and produce a dose-dependent decrease in clinical scores, indicating significant amelioration of MS-like pathologies.
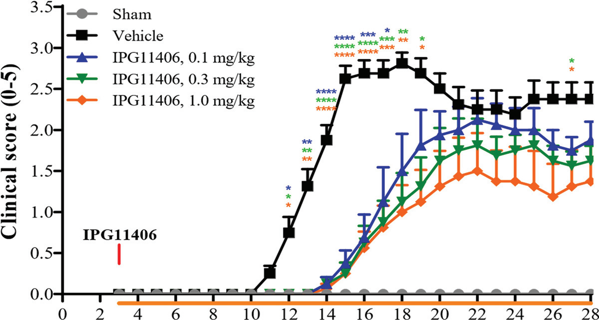
Clinical score of EAE mice for the treatment of IPG11406. IPG11406 dose dependently reduced clinical score in the murine EAE model.
• Rheumatoid Arthritis (RA): In the collagen-induced arthritis (CIA) mouse model, IPG11406-treated mice exhibited a clear, dose-dependent decrease in the clinical arthritis score and paw thickness compared to the vehicle control group.

In the collagen-induced arthritis (CIA) mouse model, IPG11406 can dose-dependently reduce clinical score, paw thickness, and the body weight.
Publications
• Xi J, Gong H, Li Z, Li Y, Wu Y, Zhang Y, Wang JF, Fan GH. Discovery of a First-in-Class GPR183 Antagonist for the Potential Treatment of Rheumatoid Arthritis. J Med Chem. 2023 Dec 14;66(23):15926-15943. doi: 10.1021/acs.jmedchem.3c01364. Epub 2023 Dec 4. PMID: 38047891.
Cpyright © 2025 Nanjing Immunophage Biomedical Co.,Ltd
All Rights Reserved. 苏ICP备2022026466号-1  苏公网安备 32011202000830号
苏公网安备 32011202000830号
